Sumalatha Peddi, Jacob R. Franklin, and C. Scott Hartley*
J. Chem. Educ. 2024, 101, 145–150
[Published version | NSF-PAR | Preprint]
Abstract
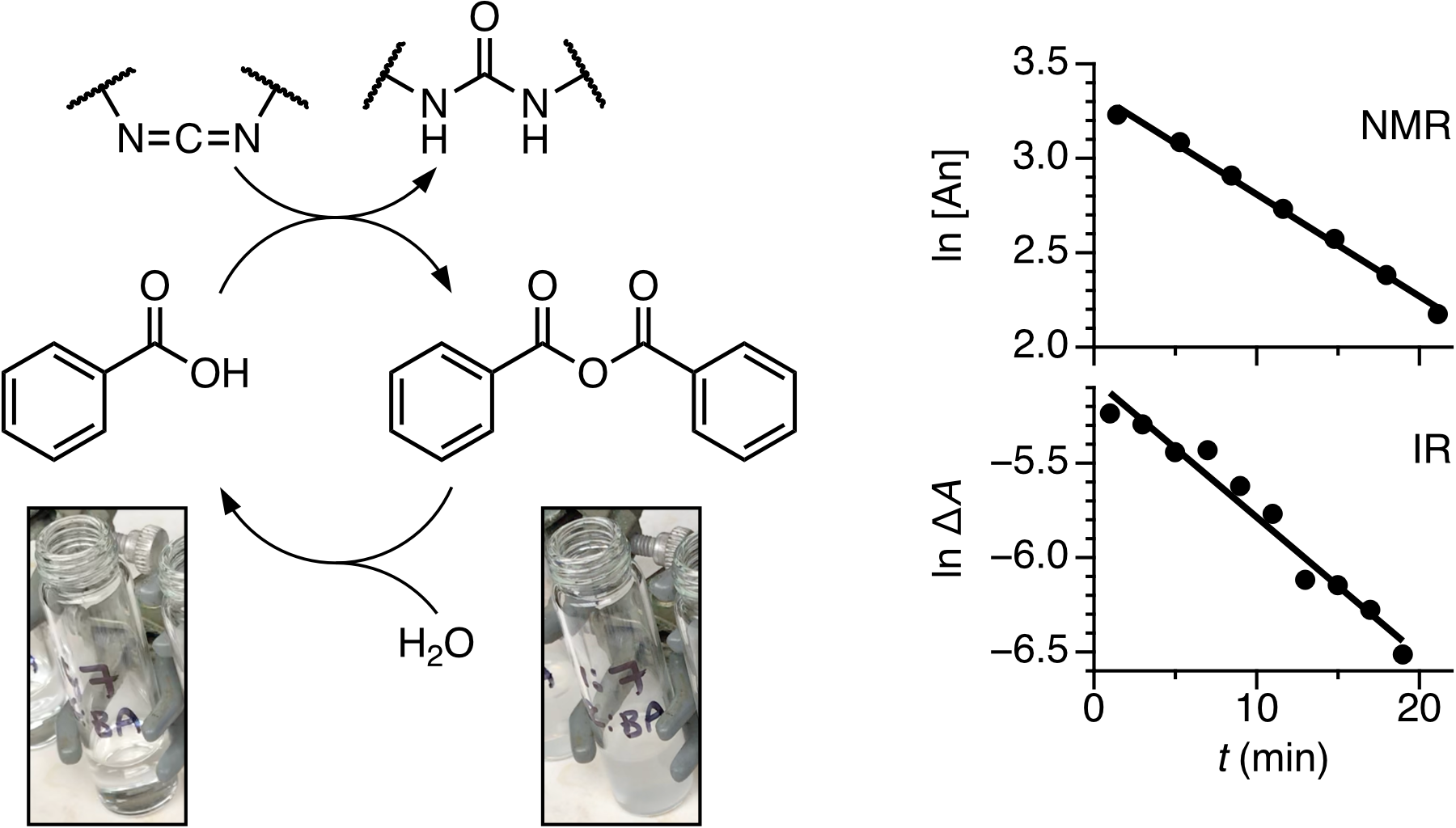
Chemical reactions that mimic the function of ATP hydrolysis in biochemistry are of current interest in nonequilibrium systems chemistry. The formation of transient bonds from these reactions can drive molecular machines or generate materials with time-dependent properties. While the behavior of these systems can be complicated, the underlying chemistry is often simple: they are therefore potentially interesting topics for undergraduate introductory organic chemistry students, combining state-of-the-art advances in systems chemistry with straightforward reactions. Here, a teaching experiment has been developed that explores the transient assembly of benzoic acid derivatives driven by carbodiimide hydration. Working in teams, students examine the formation and decomposition of anhydrides from two benzoic acids using a carbodiimide “fuel”. The students examine classic reaction kinetics of anhydride hydrolysis using two independent methods, NMR and IR spectroscopies. They then explore how the amount of carbodiimide affects the lifetimes of precipitates of benzoic anhydride as a simple example of out-of-equilibrium self-assembly.
