Sanyo M. Mathew and C. Scott Hartley*
Macromolecules 2011, 44, 8425–8432
[Published version]
Abstract
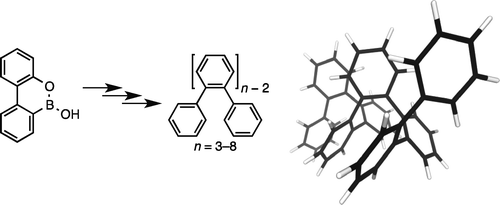
The o-phenylenes are an unusual class of conjugated polymer, defined largely by substantial steric twisting along their backbones. Consequently, they exhibit limited conjugation but also interesting conformational behavior: they have been shown to adopt well-defined helical secondary structures, both in the solid state and in solution. While several examples of functionalized o-phenylene oligomers have been reported, most of the basic properties of the parent compounds are unknown. Here we report the synthesis and characterization of the series of unsubstituted o-phenylene oligomers up to the octamer. Through a combination of NMR spectroscopy, including dynamic NMR (EXSY), and computational chemistry, we have found that these compounds adopt compact helical conformations in solution with three repeat units per turn. Although formally conjugated, the oligomers have a very short effective conjugation length of necl ≈ 4 (based on UV–vis spectra), significantly shorter than most other conjugated systems. Also, unlike other (substituted) o-phenylenes, no hypochromicity is observed in their UV–vis spectra. The fluorescence spectra of the series exhibit a systematic blue shift with increasing length. We believe this unusual property results from increased steric congestion in the longer oligomers, which are therefore less able to accommodate structural relaxation in the excited state.
