Jana Jensen, Stephan C. Grundy, Stacey Lowery Bretz, and C. Scott Hartley*
J. Chem. Educ. 2011, 88, 1133–1136
[Published version]
Abstract
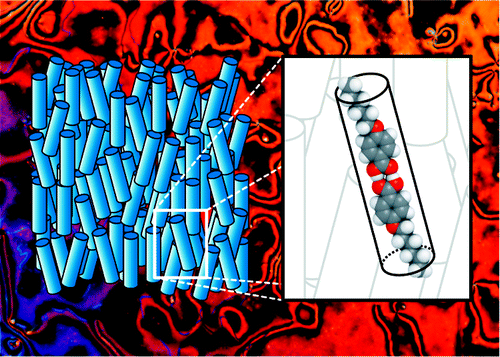
Thermotropic liquid crystal phases are ordered fluids found, for some molecules, at intermediate temperatures between the crystal and liquid states. Although technologically important, these materials typically receive little attention in the undergraduate curriculum. Here, we describe a laboratory activity for introductory organic chemistry students on the synthesis and characterization of the p-alkoxybenzoic acids. These compounds, through the formation of carboxylic acid dimers, exhibit liquid crystal phases common in rod-like (calamitic) molecules. The students are assigned different alkoxy chain lengths and synthesize the compounds through microwave-assisted nucleophilic substitution. Characterization of the phase behavior is then carried out by standard melting point techniques, differential scanning calorimetry, or polarized optical microscopy. The results for the class are pooled to allow the students to consider structure–property effects for the series. This activity allows students to explore small-molecule synthesis applied to materials chemistry and concepts of self-assembly: the benzoic acids associate through hydrogen bonding, and the resulting rod-like dimers further organize into the liquid crystal phases.
