Meng Chu, Ashley N. Scioneaux, and C. Scott Hartley*
J. Org. Chem. 2014, 79, 9009–9017
[Published version]
Abstract
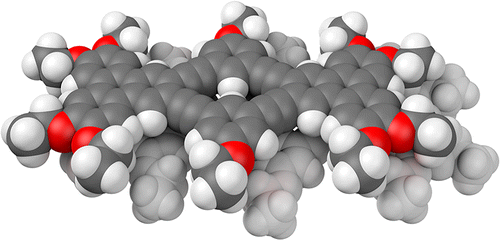
A new shape-persistent macrocycle comprising two 2,3-triphenylene moieties bridged by m-phenylene ethynylenes has been synthesized. UV–vis and fluorescence spectroscopies indicate limited interaction between the two triphenylene units. The compound, which has a pronounced oblong shape (the core measures approximately 2.2 × 1.0 nm), aggregates in CDCl3 and toluene-d8 to give stacked dimers, as indicated by the 1H NMR signals corresponding to protons on or near the core, which shift upfield with increasing concentration. These changes in NMR shielding were modeled using DFT calculations on candidate dimer geometries. The best match to the experimental data was obtained for a dimer consisting of arene–arene stacking by 3.6 Å (on average) with a displacement along the molecular long axis of 3.5–4.0 Å. This displacement is larger than can be explained by the electronic effects of aromatic stacking interactions. Instead, the minimization of steric interactions between the side chains appears to control the dimer geometry, with the alkoxy groups of one molecule sliding into registry with the gaps along the periphery of the other. Such lateral displacement (as opposed to rotation) is a direct consequence of the extended oval shape of the compound.
