Obed Dodo, Leilah Petit, Chamoni Rajawasam, C. Scott Hartley,* and Dominik Konkolewicz*
Macromolecules 2021, 54, 9860–9867
[Published version | DOE-PAGES | Raw data]
Abstract
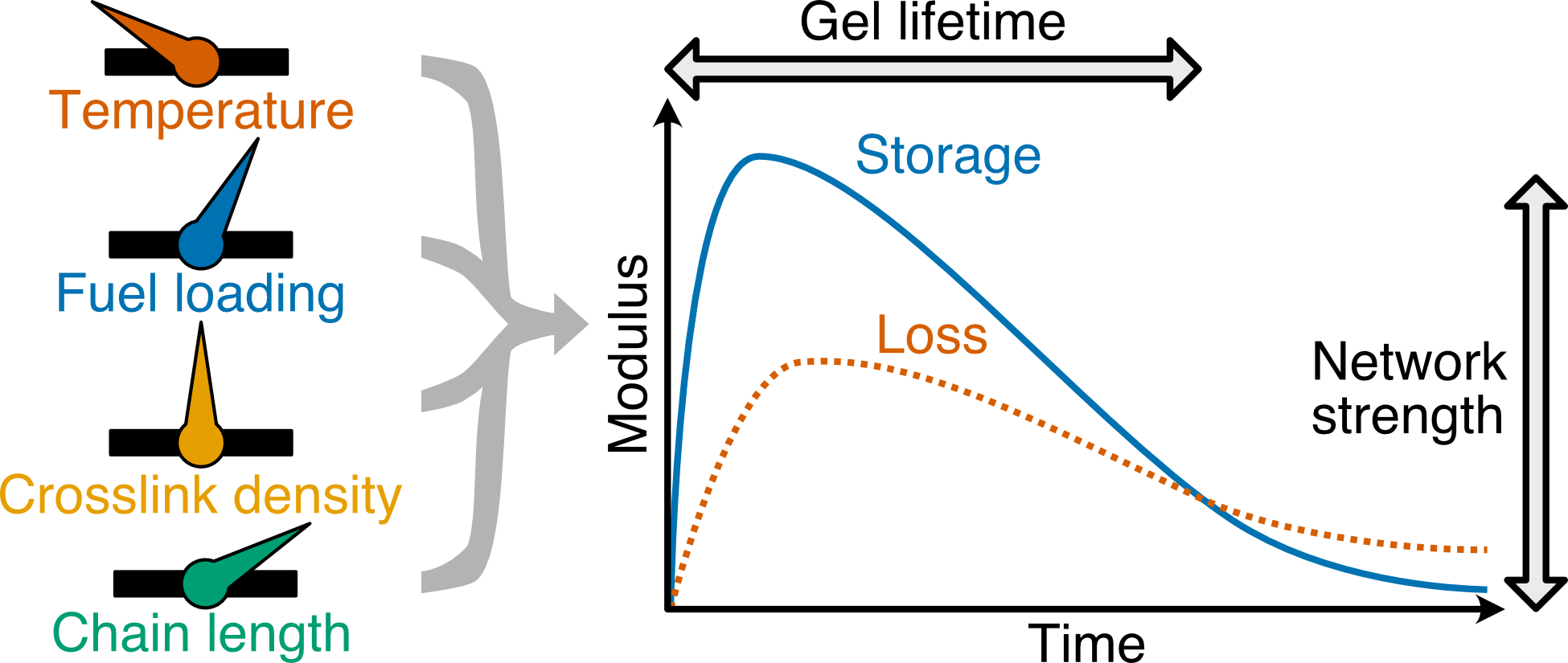
The mechanical properties of nonequilibrium polymer hydrogels obtained from the transient cross-linking of polymer chains by a chemical fuel were investigated. Aqueous polymers featuring pendant carboxylic acids were treated with a carbodiimide to give anhydride-cross-linked gels. The anhydrides spontaneously hydrolyze back to the polymer solution, and the cycle can be repeated multiple times. Oscillatory rheology was employed to study the effects of temperature, fuel concentration, chain length, and polymer composition on the storage and loss moduli of the polymeric materials as well as the time taken for the polymers to undergo decross-linking. Regardless of the temperature used, at constant carbodiimide concentration, degelation times are more sensitive to experimental temperature than are the peak storage moduli. Decross-linking times decrease with increasing temperature. As carbodiimide concentration decreases, there is a decrease in moduli and decross-linking times. Within the scope of materials studied, the polymer structure was found to have a relatively small impact on the transient properties of gel networks compared to the fuel concentration and temperature. These findings facilitate the design of tunable on-demand networks and gels.
