Sumalatha Peddi, Molly C. Bookout, Gopi Nath Vemuri, and C. Scott Hartley*
J. Org. Chem. 2022, 87, 3686–3690
[Published version | NSF-PAR | Preprint | Raw data]
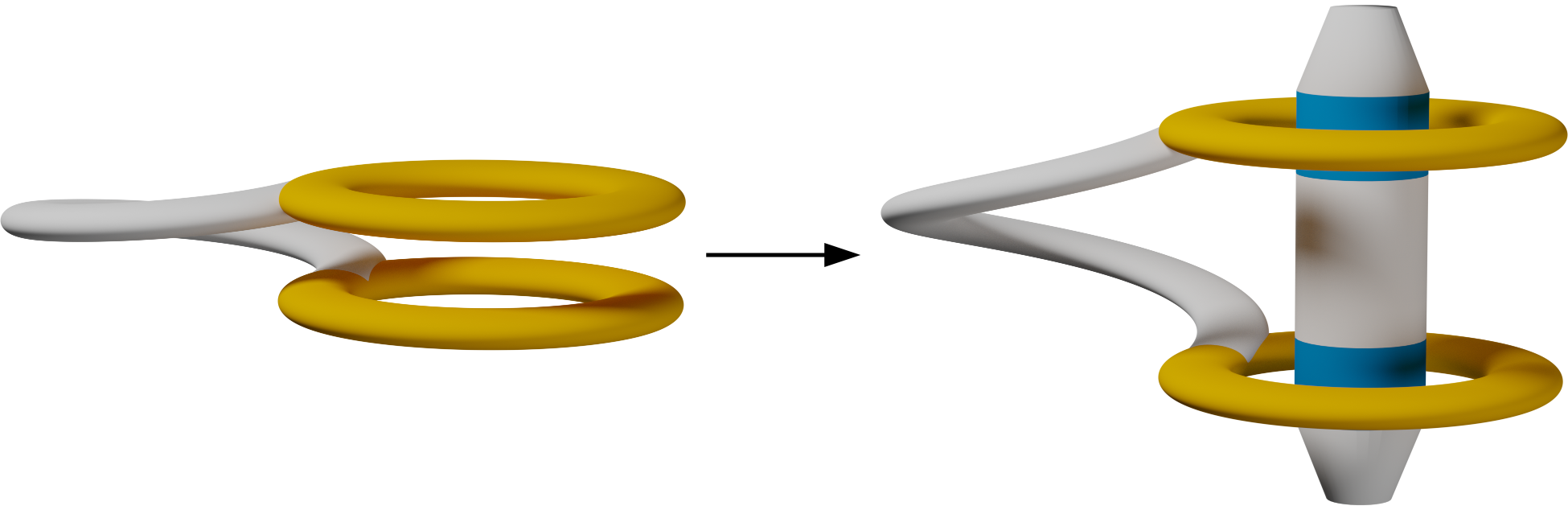
Abstract
A crown-ether-functionalized o-phenylene tetramer has been synthesized and co- assembled with monotopic and ditopic, achiral and chiral secondary ammonium ion guests. NMR spectroscopy shows that the o-phenylene forms both 1:1 and 1:2 complexes with monotopic guests while remaining well-folded. Binding of an elongated ditopic guest, however, forces the o-phenylene to misfold by pulling the terminal rings apart. A chiral ditopic guest biases the o-phenylene twist sense.
